Abstract
Background:
Hypomethylating agents (HMA) are the standard of care for higher risk MDS patients (pts). Fewer than one-half will respond to therapy for an average duration of one year, thereby emphasizing the necessity to optimize the use of these disease modifying agents. HMA clinical trials have not addressed the optimal time in the disease course to initiate treatment to maximize disease-modifying potential. The current dogma is to begin HMA therapy in all higher risk MDS pts at the time of initial diagnosis. Nevertheless, a subset of higher risk MDS pts will have adequate hematopoiesis at the time of diagnosis regardless of disease risk, and for these pts therapy may be delayed and reserved for a later time when symptomatic cytopenias develop. We investigated the impact of the timing of HMA initiation on outcomes among higher risk MDS pts presenting with adequate blood counts to discern the possible benefit of early treatment based solely on disease risk.
Methods:
We identified MDS pts with intermediate-2 and high risk IPSS (higher risk) MDS treated with HMA among the Moffitt Cancer Center database. We included patients with adequate hematopoiesis defined for the purpose of this study as platelets >50 x 109/L, Hgb > 9 g/dl and ANC > 0.5 x 109/L and being transfusion independent to exclude those pts in need of HMA treatment for cytopenias. We divided patients into 4 groups based on the time of HMA initiation (within 30, 31-60 , 61-90 and greater than 90 days from time of diagnosis). We compared baseline characteristics, best response rate to treatment using international working group criteria (IWG 2006), leukemia free survival (LFS) and overall survival (OS) among the 4 groups.
Results:
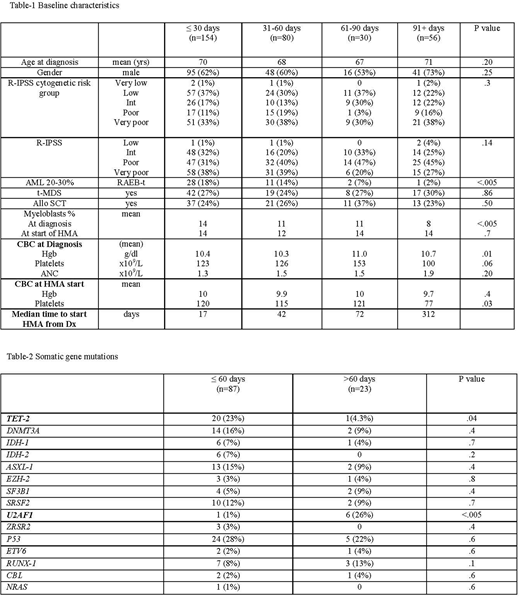
We identified 320 higher risk MDS pts with adequate hematopoiesis who were treated with HMA. Baseline characteristics for the 4 groups based upon time of HMA therapy initiation are summarized in Table-1. Pts receiving treatment within 30 days had higher marrow myeloblast percentage at time of diagnosis. There was no difference in mean blood counts at time of diagnosis between the 4 groups; however, mean platelet count was lower at time of initiating HMA therapy in patients treated after 90 days from diagnosis. Table-2 summarizes somatic gene mutation data among 110 patients tested. TET-2 mutations were more common among those treated within 60 days, whereas U2AF1 mutations were more common among those treated after 60 days from diagnosis.
The complete response rates were 21%, 26%, 23% and 7%, respectively for pts treated within 30 , 31-60 , 61-90 and greater than 90 days from time of diagnosis (p=0.046). The overall response rates (defined as hematological improvement or better) were 43%, 41%, 43% and 34%, respectively for pts treated within 30, 31-60 , 61-90 and greater than 90 days from date of diagnosis (p .70). There was no difference in mean duration of treatment with HMA among the groups with mean durations of 267, 204, 224 and 215 days, respectively, (p .35).
The median OS from the date of diagnosis was 641, 550, 979 and 806 days, respectively for pts treated within 30, 31-60, 61-90 and greater than 90 days from date of diagnosis (p .2). There was no impact of timing for HMA initiation when adjusted for Revised-IPSS risk groups in Cox regression analysis. There was no difference in OS based on HMA initiation time when adjusted for myeloblasts % or karyotype. Further, there was no difference between groups in rate of AML transformation or LFS (p =.7 and .16, respectively).
Conclusions
A delay in initiating HMA therapy in higher risk MDS pts with adequate blood counts is not associated with worsened overall survival or increased frequency of leukemia transformation. Complete response rates were higher when treatment was started earlier in the disease course, however, there was no difference in overall response rate. Close observation to initiate treatment upon development of clinically significant cytopenias appears to be a safe and equally effective strategy among pts with higher risk MDS.
Komrokji:Novartis: Honoraria, Speakers Bureau; Celgene: Honoraria, Research Funding; Novartis: Honoraria, Speakers Bureau; Novartis: Honoraria, Speakers Bureau; Novartis: Honoraria, Speakers Bureau; Celgene: Honoraria, Research Funding. Sallman:Celgene: Research Funding, Speakers Bureau. Nazha:MEI: Consultancy. Steensma:Takeda: Consultancy; Syros: Research Funding; Otsuka: Membership on an entity's Board of Directors or advisory committees; Onconova: Consultancy; Novartis: Membership on an entity's Board of Directors or advisory committees; Kura: Research Funding; Janssen: Consultancy, Research Funding; H3 Biosciences: Research Funding; Celgene: Research Funding; Amphivena: Membership on an entity's Board of Directors or advisory committees; Acceleron: Consultancy. Roboz:AbbVie: Consultancy; Daiichi Sankyo: Consultancy; Jazz Pharmaceuticals: Consultancy; Cellectis: Research Funding; Novartis: Consultancy; Celltrion: Consultancy; Astex Pharmaceuticals: Consultancy; Sandoz: Consultancy; Celgene Corporation: Consultancy; Otsuka: Consultancy; Janssen Pharmaceuticals: Consultancy; Aphivena Therapeutics: Consultancy; Daiichi Sankyo: Consultancy; Orsenix: Consultancy; Bayer: Consultancy; Aphivena Therapeutics: Consultancy; Argenx: Consultancy; Orsenix: Consultancy; Novartis: Consultancy; Jazz Pharmaceuticals: Consultancy; Eisai: Consultancy; Celgene Corporation: Consultancy; AbbVie: Consultancy; Roche/Genentech: Consultancy; Cellectis: Research Funding; Otsuka: Consultancy; Bayer: Consultancy; Pfizer: Consultancy; Astex Pharmaceuticals: Consultancy; Argenx: Consultancy; Sandoz: Consultancy; Celltrion: Consultancy; Janssen Pharmaceuticals: Consultancy; Pfizer: Consultancy; Eisai: Consultancy; Roche/Genentech: Consultancy. Sekeres:Celgene: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Opsona: Membership on an entity's Board of Directors or advisory committees; Opsona: Membership on an entity's Board of Directors or advisory committees. List:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal